Author: Brian N. Lundstrom, M.D., Ph.D., epileptologist Mayo Clinic
This blog post is sponsored by Mayo Clinic.
When people have recurrent seizures, they are diagnosed with epilepsy, which is defined as a tendency to have seizures. Epilepsy is first treated with medications. However, about one-third of people continue to have seizures despite trying multiple anti-seizure medications. If these seizures are focal seizures, then it could be that removing an appropriate portion of the brain by surgery would lead to seizure freedom. The evaluation for this possibility is extensive and was discussed in a previous blog. If these seizures are generalized onset seizures or surgical resection is not a possibility, then brain stimulation may be an appropriate treatment approach for these drug-resistant seizures.
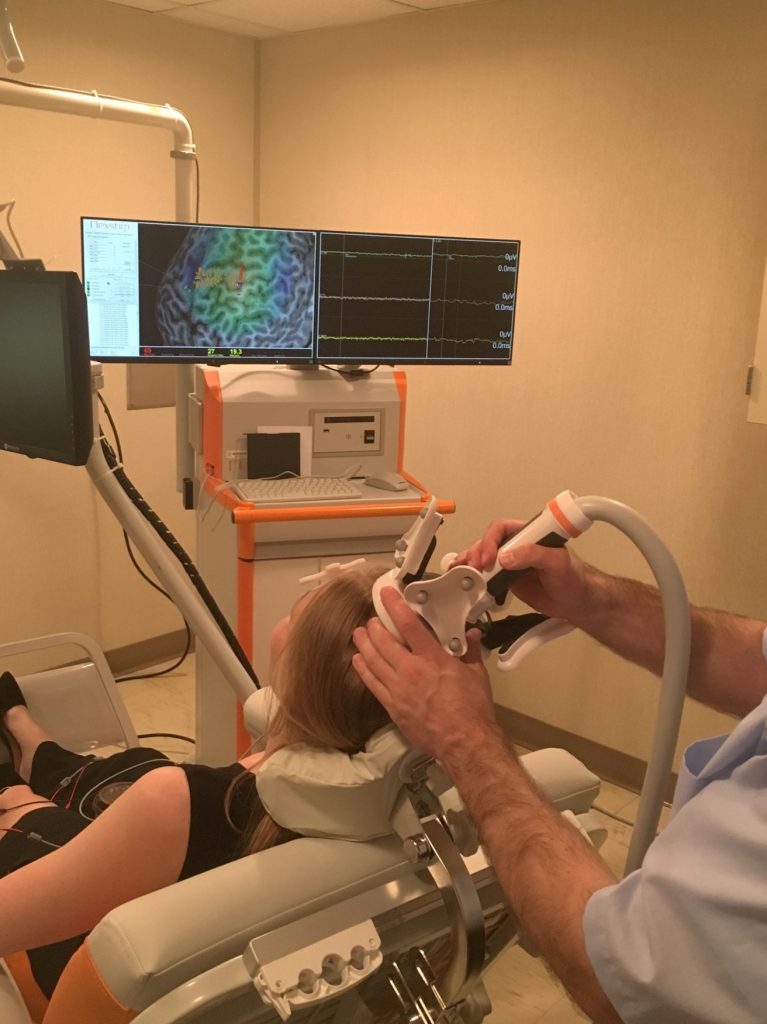
In the past 20 years or so, there have been considerable developments in the area of brain stimulation for epilepsy. In general, brain stimulation can be categorized as either invasive stimulation or noninvasive stimulation. Invasive stimulation involves electrodes that are permanently placed in the body to provide electrical stimulation. Right now, there are three Food and Drug Administration (FDA)-approved options for invasive brain stimulation: vagus nerve stimulation (VNS), responsive neurostimulation (RNS) and deep brain stimulation of the anterior nucleus of the thalamus (DBS-ANT). At present, no noninvasive brain stimulation approaches are FDA approved.
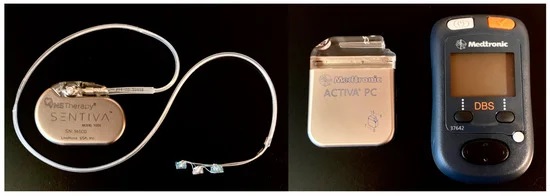
For invasive brain stimulation approaches, a device must also be implanted. This device is sometimes called an implantable pulse generator (IPG) and is where the electrical pulses are generated. The IPG is similar in size to a deck of cards and typically placed either in the chest or embedded in the skull. Depending on the approach, sometimes a rechargeable IPG is an option. Otherwise, the device is typically replaced approximately every five years. In general, invasive brain stimulation does not lead to seizure freedom and is considered a palliative approach. However, evidence does suggest that its efficacy improves over time.
VNS was FDA approved in 1997 and involves stimulation of the vagus nerve. The name for this large nerve means “wandering” in Latin, and stimulation of this nerve leads to decreased frequency of seizures by modulating neurotransmitters in the brain. Implantation of the VNS device does not involve brain surgery but rather neck surgery. Electrodes are implanted around the left vagus nerve, and the IPG is placed in the upper left portion of the chest. The most typical side effect is that of hoarseness and coughing. For this reason, the level of stimulation is typically increased over one to two months. Often considered the safest form of invasive stimulation, VNS often leads to an approximate 30% to 40% reduction in seizure frequency by the end of the first year. It is often used for generalized onset seizures as well as focal seizures and is FDA approved for patients as young as 4 years of age.
RNS was FDA approved in 2013 for treatment of focal seizures and is currently approved for patients 18 years and older. For this approach, the location in the brain where focal seizures originate needs to be determined. Sometimes this involves invasive EEG recordings, in which temporary electrodes are placed in the brain to record seizure activity. Once the seizure onset zone is localized, the RNS electrodes can be permanently implanted to sense seizure activity and stimulate the brain. The RNS IPG is placed in the skull. Electrical stimulation is provided intermittently when RNS electrodes sense seizure-related brain activity. The way stimulation is delivered is optimized over months, and it can take six to 12 months before notable benefits are seen. Typically, seizure frequency is reduced by half at the end of the first year. This benefit slowly improves in the following years.
DBS has long been performed as treatment for Parkinson’s disease or tremor. However, in 2018 DBS was approved for treatment of focal seizures by stimulating the anterior nucleus of the thalamus, which is located at the front of the thalamus. Thalamus is a Greek word that means “bedroom,” and this structure is located near the middle of the brain with a left and right side. The thalamus is a relay center. By stimulating the thalamus, the idea is that seizure activity is stopped before it spreads to the rest of the brain. Because two electrodes are placed in the front part of the thalamus for each patient, it is not necessary to know precisely where seizures start. Like VNS, the IPG for DBS is typically placed in the upper part of the chest. Overall seizure reduction results are similar to those of RNS.
There are noninvasive methods that have been used to treat seizures. However, none of them is currently approved by the FDA for treatment of epilepsy. Noninvasive methods include transcranial magnetic stimulation (TMS) and transcranial direct current stimulation (tDCS) and are typically considered low-risk procedures. TMS uses an electromagnet to create a changing magnetic field that then induces a brief electrical pulse in the brain. By using a magnetic field to produce the electrical pulses, relatively large electrical currents can be produced without causing scalp pain. However, it requires relatively expensive technology to create the TMS pulses. In contrast, tDCS uses very weak electrical fields applied to the scalp to create electrical fields in the brain. This form of stimulation can be produced using inexpensive technology and is the basis of a number of direct-to-consumer marketed devices. Both TMS and tDCS have been studied as forms of treatment for epilepsy with promising results.
About the author:
Brian N. Lundstrom, M.D., Ph.D., is a neurologist and epileptologist at Mayo Clinic in Rochester, Minnesota, who works with brain stimulation devices. His clinical and research interests include invasive and noninvasive brain stimulation, the development of EEG biomarkers, and computational neuroscience applied to dysfunction of cortical excitability.
About Mayo Clinic:
Mayo Clinic wants to make sure you feel empowered to make the right decisions for your care. The goals of treatment include no seizures, side effects or lifestyle limitations. You are at the center for your care, and Mayo Clinic’s team of multidisciplinary experts is ready to provide a personalized treatment plan for you. Beyond medications, Mayo’s teams of clinician researchers are on the cutting edge of new treatment options for people with seizure disorders. Advanced diagnostic technology help pinpoint the root cause of your seizures and help Mayo’s teams identify your best treatment options. Mayo’s top-ranked teams evaluate and treat more than 8,000 adult and pediatric patients with epilepsy each year. For more information or to request an appointment please visit mayoclinic.org/epilepsy.